There has been a lot of miscommunication and misinformation surrounding the production, distribution, and administering of the COVID-19 vaccines. Locally, the general public has little information on the same which has led to the propagation of inaccurate information causing unnecessary unrest. In view of that, I went on a fact-finding mission with experts in the field to ensure that correct information is being passed around so that any decision made in terms of uptake of the vaccine is made from an informed point.
Here is our conversation with Shem Mutuiri, a Medical Microbiologist and a Lecturer of Microbiology, Virology, and Immunology from Kenyatta University.
What are vaccines?
Vaccines are substances, usually whole organisms or proteins that make the immune system (white blood cells) “aware” of the existence of a pathogen and trigger the immune cells to produce antibodies (also proteins) that attach to the antigen if in the future the pathogen or its antigens enter the body.
How do vaccines work?
It is a complex process that involves immune cells (white blood cells) known as Antigen Presenting Cells (APCs) and T cells. the two types of T cells are CD4 helper cells & CD8 cytotoxic cells. APCs capture any invading pathogen then interact with CD4 helper cells that help the CD8 cells to kill the pathogen and also help the Plasma Cells (B cells) to identify the pathogen and produce antibodies to neutralize the pathogen.
What are the different types of vaccines?
There several different types of Vaccines which include but not limited to;
Live vaccines-
These usually contain the whole organism that has been attenuated (made not to cause disease). This then interacts with the immune cells and makes the body remember and be able to fight off any subsequent infection from the same pathogen. However, these types of vaccines can revert back to pathogenicity which means that they can cause disease, especially in people with low immunity. E.g. the Oral Poliovirus vaccine (OPV). The advantages of live vaccines are; they cheaper, easier, and less time coming to produce and they ensure that the immune system fights back with humoral (production of antibodies by the B cells) and cellular immunity involving all white blood cells and memory cells.
Killed/ Inactivated Vaccines
These are produced by killing the disease-causing microorganism with either chemicals, heat or radiation, with end result being that the vaccines themselves are safer and don’t mutate and end up causing disease. E.g the Polio Inactivated Poliovirus Vaccine (IPV). These vaccines are also easier to store and transport with most even withstanding very high climatic/ temperature conditions (especially in the tropics).
Subunit Vaccines
These vaccines are produced using a small part of the whole organism thus the term “subunit”. However, the hard part of their production and successful use lies in the identification of which part of the entire microbe will be best in stimulating the immune system to fight off the organism in future infections. They are smaller than the live or killed vaccines thereby have fewer side effects. The process of identifying the “Best” small piece (subunit) of the organism, however, can be hard scientifically speaking, and time-consuming too. E.g. Hepatitis B virus vaccine.
Toxoid Vaccines
Some bacteria like Mycobacterium tetani which causes tetanus cause disease by producing poisons known as toxins. These toxins either have a direct effect on the infected host’s cell, tissues and organs thereby causing disease. In addition, they can use the immune system to react harshly with this explosive immune reaction being the end resulting disease. The toxins are inactivated by the use of chemicals such as formaldehyde and sterilized water solution and then they can be injected into humans as safe toxoids e.g. diphtheria and tetanus vaccines.
Conjugate Vaccines:
Certain bacteria contain an outer coating made up of sugar molecules called the peptidoglycan layer. This coating hides the bacterium’s antigens so that the immature immune systems of infants and younger children can’t recognize or respond to them. To get around this problem scientists develop what are known as Conjugate vaccines. This special type of vaccine contains the sugar coating which is conjugated (linked or attached) to a known antigen or toxin that the immune system can then recognize and react to both the toxin and the polysaccharide (especially in infants and children; thus the choice of the antigen for linking to the sugarcoating of the bacteria is crucial). E.g. Haemophilus influenzae type B (Hib) is a conjugate vaccine.
DNA/mRNA Vaccines:
After the discovery of a new microorganism or after research into a microorganism of interest has been done, one of the researches that can be done involves the analysis of the genes. Genes are the small basic units that are transferred from the parents to the offspring and are made up of Nucleic acids known as Deoxyribonucleic Acid (DNA) for animals including humans and microorganisms. Some genes code for or result in the production of proteins through the production of Ribonucleic acid (RNA) which is another type of nucleic acid. However, some viruses can be made up of RNA alone which codes for the viral proteins and that’s why they are only able to reproduce only inside a cell and can’t reproduce on their own. From a microbe that has already been analyzed, DNA can be created that resembles that of the microorganism itself or that codes for proteins similar to those produced by the bacteria itself. The DNA gets inside the human cell and instructs it to produce antigens (proteins) similar to those of the disease-causing microorganism itself. Cells with this DNA then end up displaying these antigens on the surface or releasing them thereby ensuring the immune system reacts to them as it would the microbial infection itself. In the case of mRNA vaccines, a similar reaction occurs when the mRNA enters into and directly goes to ensure that the cells produce the proteins then end up displaying these antigens on the surface or releasing them thereby ensuring the immune system reacts to them as it would the microbial infection itself. The DNA/ mRNA vaccine couldn’t cause the disease because it wouldn’t contain the microbe, just copies of a few of its genes. In addition, mRNA codes for the production of a few proteins of the microorganism usually the virus vaccines but not the whole microbe. They are relatively easy to produce, less expensive than most other vaccines due to current advances in science, and easy to administer with a needle syringe or pressured needleless device (to shoot microscopic gold particles coated with DNA) as their “small” nature thus the vaccine solutions enter the body and cells more easily. E.g. COVID19, influenza and herpes vaccines.
Recombinant Vector Vaccines
These vaccines are similar to DNA vaccines but the DNA, in this case, is carried by using an attenuated virus or bacterium to introduce microbial DNA to cells of the body. In addition, there can be the use of a Virus or Bacterium that only causes disease in another orgasm. “Vector” refers to the virus or bacterium that is used as the carrier. In nature, viruses latch on to cells and inject their genetic material into them. In the lab, scientists have taken advantage of this process. They have figured out how to take the roomy genomes of certain harmless or attenuated viruses and insert portions of the genetic material from other microbes into them. The carrier viruses then ferry that microbial DNA to cells. Recombinant vector vaccines closely mimic a natural infection and therefore do a good job of stimulating the immune system. Attenuated bacteria also can be used as vectors. In this case, the inserted genetic material causes the bacteria/virus to display the antigens of other microbes on its surface. In effect, the harmless bacterium mimics a harmful microbe, provoking an immune response. E.g. The Astrazeneca-Oxford vaccine uses The Adenovirus from Chimpanzee that delivers the mRNA to the cells but does not replicate. Other examples are several HIV vaccines, rabies, and measles vaccines.
How long is the typical time taken to develop a vaccine?
According to the CDC, typically, there are three phases of clinical trials. Vaccines that are being developed for children are first tested in adults. FDA sets guidelines for the three phases of clinical trials to ensure the safety of the volunteers.
Phase 1 clinical trials focus on safety and include 20–100 healthy volunteers. In Phase 1, scientists begin to learn how the size of the dose may be related to side effects. If possible at this early stage, scientists also try to learn how effective the vaccine may be.
If no serious side effects are found in Phase 1, next is Phase 2, which involves several hundred volunteers. This phase includes studies that may provide additional information on common short-term side effects and how the size of the dose relates to immune response.
In Phase 3 studies, hundreds or thousands of volunteers participate. Vaccinated people are compared with people who have received a placebo or another vaccine so researchers can learn more about the test vaccine’s safety and effectiveness and identify common side effects.
The trials and all other data must show that the vaccine’s benefits outweigh the potential risks (side effects) for people who will be recommended to receive the vaccine. Only if a vaccine’s benefits are found to outweigh its potential risks does the FDA grant a license for the vaccine, allowing it to be used by the public.
The last trial,(Sometimes Not numbered a Phase Four) Rollout Phase or Phase Four or Vaccine efficiency Monitoring Phase
After vaccines are licensed, they are monitored closely as people begin using them. The purpose of monitoring is to watch for adverse events (possible side effects). Monitoring a vaccine after it is licensed helps ensure that the benefits continue to outweigh the risks for people who receive the vaccine.
Monitoring is essential for two reasons. First, even large clinical trials may not be big enough to reveal side effects that do not happen very often. For example, some side effects may only happen in 1 in 100,000 or 1 in 500,000 people.
Second, vaccine trials may not include groups who might have different types of side effects or who might have a higher risk of side effects than the volunteers who got the vaccine during clinical trials. Examples of these groups include people with chronic medical conditions and pregnant women.
If a link is found between a possible side effect and a vaccine, public health officials take appropriate action by first weighing the benefits of the vaccine against its risks to determine if recommendations for using the vaccine should change.
How long is the typical time taken to develop a vaccine?
Successful Vaccine development times have changed over history with no definite times ranges from more than 60 years for Poliovirus to 35 years and counting for HIV to probably less than a year for the Edward Jenner smallpox vaccine.
What causes COVID-19?
COVID-19 is a respiratory disease is caused by Severe Acute Respiratory Syndrome Coronavirus 2 (SARS-CoV-2). The Virus is of zoonotic origin thus is referred to as a zoonotically transmitted virus and the disease is referred to as a Zoonotic disease. According to WHO and ICTV, Viruses, and the diseases they cause, often have different names. For example, HIV is the virus that causes AIDS. People often know the name of a disease, but not the name of the virus that causes it. COVID-19 disease is characterized by several and diverse symptoms in different people ranging from a mild illness to moderate illness that recovers without hospitalization to Serious symptoms such as difficulty in breathing or shortness of breath, systemic blood clots, hypoxia (low oxygen levels in the blood), chest pain or pressure, loss of speech or movement. The most common symptoms include fever, dry cough, and tiredness. There has been the emergence of new strains and also the viral-host interaction that has resulted in the reporting of these less common symptoms in higher numbers; general body aches and pains, sore throat, diarrhea, conjunctivitis, headache, loss of taste or smell, a rash on the skin, or discoloration of fingers or toes. Severe acute respiratory syndrome coronavirus 2 is a member of a large family of viruses called coronaviruses. These viruses can infect people and some animals. The coronaviruses infecting humans comprise six species with one species subdivided into two different strains, making seven strains of human coronaviruses in total.
Can COVID-19 infect animals?
Yes. According to American Veterinary Medicine Association, Since the initial outbreak of SARS-CoV-2 and COVID-19, there have been reports of animals becoming infected with the virus.
Can a healthy person with no underlying medical condition die from COVID-19?
Sadly, A big Yes. There have been several cases of people without underlying conditions dying from COVID-19. One major concern for the medical practitioners dealing with infection of people below the age of 50, is that some may have underlying conditions especially heart disease that is undiagnosed at the time of infection. Other predisposing factors especially for patients hospitalized or dying from Severe acute respiratory syndrome (SARS) which is the acute form of the disease from the virus has been shown to even be a history of allergies, with others having cytokine storms which is the release of the cytokines (messengers found in the blood) which might be genetic. The virus is known to infect cells through the ACE2 receptor found in several organs, which means that if one has a problem, for example, kidney disease or TB infection of the lungs that was undetected the virus can infect the kidney, resulting if “faster” total kidney failure or Pneumonia and eventual death. Deaths resulting from hypoxia, which is low oxygen in blood have also been shown to cut across all groups including those without any underlying conditions and including people under 50 years. Blood clotting and blood clotting correlated disorders have been higher in the elderly and people with other underlying conditions like diabetes but nonetheless, there have been cases in young people especially home caregivers, indicating that this might be linked to viral load levels or reinfections as one spends more time with the patient who might be a relative making one not to observe the Ministry of Health (MOH) guidelines. In addition to this, the virus has had unique cases where some people have remained positive (and infectious) for several days, suggesting that the 14 days quarantine period, may not be enough (https://www.aa.com.tr/en/asia-pacific/14-day-quarantine-not-enough-for-covid-19-patients/1948886).
So to all the people out there saying that they are healthy people with no underlying medical condition and thus can’t die from COVID-19, should only support this with Full body checks, Full blood (haemogram) tests, Organ scans, cancer tests, and heart & lung disease tests to confirm this. But even then issues like drug abuse e.g. alcohol binge drinking have been linked to sudden death resulting from Pneumonia that is not of infectious origin.
As of 12th April 2021;
Globally infections are at= 136M
Globally Recoveries are at=77.5M
Globally, death rates are at= 2.94M
Kenya infections are at= 146,486
Kenyan recoveries are at= 99,095
Kenya, death rates are at=2,348
The current problem we have in COVID-19 Infection rates is a scientific one, in that from the onset, data collection for reinfections has not been done. In addition to this, for the death rates, even patients who have had cancer or other fatal conditions and contracted the virus be it at home as a community infection or at the hospital as a nosocomial infection (these are the disease that one acquires while under hospitalization) their cause of death is recorded as COVID-19. One of the leading cause of death for persons hospitalize especially those in ICU is a bacterial illness caused by a bacteria known as Staphylococcus aureus with one strain of it known as Methicillin-Resistant Staphylococcus aureus causing death in 2017, to an estimated 119,247 S. aureus bloodstream infections with 19,832 associated deaths occurred MMWR Early Release on the MMWR website (https://www.cdc.gov/mmwr).
Who approves the use of developed vaccines?
The process of Vaccine development usually varies from region to region and from country to country. In Kenya for example, the development of the prototype vaccine starts with the institution where the development step and phase 1 trials will be done. Institutes like Kenyatta University, Institute of Primate Research (here one can even do trials with none human primates like baboons or sykes), University of Nairobi, Jomo Kenyatta University of Agriculture and Technology, National Museums of Kenya among others have Institutional Research and Ethics committees that give approvals for those undertaking research in their institutions. Other Institutions like Kenya Medical Research Institute has an Ethical review committee with a national mandate. In addition to this, research is done with the issuance of research permits from organizations such as The National Commission for Science, Technology & Innovation. In Kenya, new drugs and vaccine, even if they have gone through Phase 3 or Phase 4 clinical trials have to be approved by the Pharmacy and Poisons Board, and batches have to be tested for quality by the Kenya Bureau of Standards (KEBS) and labeled with the KEBS sign. However, in special cases as that of the COVID19 approval by the WHO, EMA, GAVI, FDA may mean that countries like Kenya can use them (for emergency use) without “repeating” the approval process and the Ministry of Health can approve them directly for clinical use. In the US, regulations, and permits for Vaccine production and use are given by The U.S. Food and Drug Administration’s (FDA’s) Center for Biologics Evaluation and Research (CBER). In the approval process, the following is done in a step by step manner;
- An Investigational New Drug application
- Pre-licensure vaccine clinical trials
- A Biologics License Application (BLA)
- Inspection of the manufacturing facility
- Presentation of findings to FDA’s Vaccines and Related Biological Products Advisory Committee (VRBPAC)
- Usability testing of product labeling
- After approving a vaccine, FDA continues to oversee its production to ensure continuing safety. Monitoring of the vaccine and of production activities, including periodic facility inspections, must continue as long as the manufacturer holds a license for the vaccine product.
FDA can require a manufacturer submit the results of their own tests for potency, safety, and purity for each vaccine lot. FDA can require each manufacturer submit samples of each vaccine lot for testing.
If a vaccine is developed in under one year is it safe for use?
Throughout recorded human history several vaccines have been developed and approved for use with different timelines and some organisms proving too “tricky” to keep up with especially those that mutate very quickly especially the viruses. It is also key to note that some of the vaccines in present-day use were developed and approved for use when current-day protocols, testing equipment, animal models, molecular techniques were not available. As such it has been shown over time, how some adverse effects resulted from the growth of the viruses in eggs and administration to people allergic to eggs necessitating the change of the manufacturing process. Therefore with this in mind and looking at the issue of whether the benefits outweigh the potential risks, I would very strongly state that vaccines can be produced in less than a year and still be safe. However, cautions needs to be taken especially for vaccines using “newer” technologies and Viral vectors as it has been shown it can have far-reaching issues. This was shown in the STEP HIV vaccine trial, which used a rAd5-based (Recombinant Adenovirus 5) vaccine developed by Merck. The vaccine failed to protect Ad5-seronegative individuals against infection and may even have enhanced infection in vaccines with prior immunity to adenoviruses, which means those who got the vaccine became more prone to acquiring the virus than those who weren’t vaccinated due to their immune reaction to the vector virus used. (https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2234358/)
If a vaccine is developed in Africa like the Johnsons and Johnsons (J&J) vaccine, who approves its use by Africans? Can it be used in Europe and America?
What does the label ‘For use in Africa only’ in medicine and vaccine bottles mean?
Approval for the J&J vaccine has proven controversial with some media houses reporting that it was developed and used in Africa without previous approval in the US. However, the released data, shows that the study participants were of almost all racial backgrounds. The statement indicated that in a trial with 43,783 participants across the US, Latin America, and South Africa, the vaccine prevented about 66% of Covid-19 cases. The company found evidence of some immunity in participants two weeks after they got the shot, which appeared to strengthen over time. It also protected against 85% of moderate-to-severe Covid-19 cases – the kinds that would lead someone to seek medical attention – and has so far presented complete protection from death due to Covid-19. However, these results varied depending on where the study participants were. In the US, the jab prevented 72% of COVID-19 cases a month later; in South Africa, it was only 57%. In Latin America, it was 66%. The ‘for use in Africa only’ label is a controversial tag given to some drugs and vaccines whose target market is African. The reason for this may be several factors including; funding received from philanthropic individuals, Government funding agencies like USAID, and other donor agencies to manufacture drugs/vaccines to “help” people in Africa who may not afford the drug or vaccine at its original cost. The other factor may be that the vaccine or drug is produced with the African market in mind where hot temperatures are expected and lack of refrigeration facilities thus the vaccine or drug is made in a way to withstand the harsh conditions. The last reason, which may not be the case for any known drug/vaccine is a scientific one where the drug is made in a way to target a certain genetic condition only found in Africans or Black Americans. There are several diseases, conditions, or reactions to drugs or vaccines that have been shown to have a racial correlation but in usual circumstances, such products are pulled off the market.
What is the difference in mechanism of action between the AstraZeneca and Sputnik V vaccine?
The vaccine both work by inducing both the Humoral immunity that involves the production of antibodies by plasma cells after differentiation from their immature state as B lymphocytes. In addition, they stimulate Cellular immunity that results in the killing of cells expressing the foreign virus antigens.
According to GAVI, (Global Alliance for Vaccines), the type of Vaccine design used may have a tangible effect on the vaccine efficacy. Both the AstraZeneca and Russian Sputnik V are vector-based vaccines. For the Astrazeneca vaccine, it’s not yet known why the reduced-dose regimen of the vaccine showed better efficacy in trials, but it could be down to the viral vector. One of the shortcomings of viral vector vaccines is that not only do the vaccinated persons develop immunity coronavirus’s spike protein being expressed once the virus injects the viral RNA to the cells of the vaccinee and the resultant expression of the protein but also, there is the development of the immunity to the viral vector vaccine. Therefore subsequent boosting may mean that the person will react even more to the viral vector thereby destroying some of the injected booster doses. However, a lower first dose might not allow for a strong anti-vector immune response to develop, which could leave the booster dose unscathed and lead to greater overall efficacy. If it turns out that this is the case, then future work will need to establish the optimum dosing regimen for generating the strongest immune response.
The Russian Sputnik V vaccine developers acknowledge that immunity to the viral vector could be a problem but comes up with a different solution. It uses two different human adenoviruses – Ad26 and Ad5 (out of the 50 that affect humans) – for its two vaccine doses. This heterologous (or hybrid) vaccine, with different vectors for prime and booster vaccinations, is less likely to have one jab generate an immune response against the viral vector that then interferes with the other. The vaccine is therefore less likely to have reduced efficacy.
Source: Nature Paper: https://www.nature.com/articles/s41541-021-00292-w
What is a booster dose?
The Merrian-Webster thesaurus defines a booster dose as a supplementary dose of a therapeutic agent designed to increase the effectiveness of one or more previously administered doses. This therapeutic agent may be a vaccine like the Yellow Fever Virus or the Oral Polio vaccine give when traveling to an endemic country or region to provide higher protection. The therapeutic agent maybe even a drug like prednisolone which is a steroid to boost the effects if the patient who taken a single dose does not experience the required clinical effects.
What happens if I take the first dose of AstraZeneca and after 8 weeks I choose to take the booster dose for Sputnik V?
There are currently no published data on vaccine interactions for the newly developed vaccine. However, it is key to note that approved childhood vaccines are given at separate time for two main reasons; to ensure that each vaccine is provided with the necessary time to interact with the immune system and to ensure that there is no interaction between the two vaccines where one may reduce the efficacy of the other vaccine. However, noting that the second dose will be 8 weeks apart and the two vaccines have different modes of action, then the possibility of interference may not occur. Sadly also, from an immunological point of view, note that the second vaccine is supposed to enhance or boost the first dose by causing a more enhanced response following re-exposure to the SAME antigen utilizing the same pathway, and as such this may not be achieved if one mixes the types of vaccines. Countries like Australia have ordered more than 5 types of Vaccines though there has been no official Australian government report on this and the planned rolling out of the Vaccines. This unique array of orders and the higher possibility that individuals in Australia may receive different vaccines may provide a better answer to this from the data from the country (read continent).
What happens if one does not take the 2nd dose on time and delays for say a week or two?
The data from the clinical trial for AstraZeneca gives a time range of between 8-12 weeks for one to get the vaccine. A dosing “mistake” in one of the AstraZeneca-Oxford vaccine clinical trials, where volunteers received the first vaccine in less amount (half dose) and the second booster vaccine as a full dose, showed that the efficacy of a half dose followed by a standard dose was 90% compared to full doses vaccine efficacy at 59%. In addition, delays to 12 weeks or even more seemed to have a positive effect. Nonetheless, more data, especially globally for all who have received the vaccine will be needed to give true scientific implications. A shortage of supplies may also mean that globally many people have to wait longer for the booster vaccine. Let’s hope this will not be the case, but if it does happen, it might as well provide the scientific community with the much-needed “sweet” data to unravel this mystery of the vaccine-vaccine-boost technique.
What happens if one skips dose 2 altogether?
You might be lucky enough to not get infected if the people vaccinated are enough to result in a phenomenon know as Herd Immunity. Herd immunity is defined as the resistance to the spread of an infectious disease within a population that is based on pre-existing immunity of a high proportion of individuals as a result of previous infection or vaccination. For COVID-19, this has been estimated to be 60% of the population. The COVAX Consortium is currently ensuring the distribution of the Vaccine with 172 economies participating. This is the very first global initiative aimed at working with all vaccine manufacturers to provide countries worldwide equitable access to safe and effective vaccines, once they are licensed and approved. COVAX currently has the world’s largest and most diverse COVID-19 vaccine portfolio – including nine candidate vaccines, with a further nine under evaluation and conversations underway with other major producers. Sadly the expected numbers of vaccine vials to be received in Kenya indicates a possible 40% coverage which is below the threshold. However, the natural infection may provide protection for non-vaccinated-asymptomatic-persons. Moreover, whether vaccinated or not, people should ensure that they stick to the Ministry of Health guidelines & protocols like washing of hands, avoiding gathering, sanitizing hands and surfaces where possible. The vaccines should be seen as one way of killing this invasive rat known as SARS-Cov-2 (On a light note, I hope this statement doesn’t sound like animal cruelty and if it does, I hereby give my heartfelt apology)
If I have previously suffered from COVID-19, should I still take the vaccine, and if so, why?
If after your last infection you have stayed for more than 12 weeks without reinfection, you can take the vaccine. COVID19 Vaccinations is voluntary and that’s why I have not used the term, “you should”. However, data trickling in from various publications indicate that naturally infected persons have a level of protection and if re-infected they have been noted to have a level of immune-boosting. However with scanty data, and they’re already being a number of strains of the virus, the infection may not be equaled to protection from disease. Previously infected persons may decide not to take the vaccines and this may result in the positive effect of the scarcely available vaccine vials being enough for everyone who needs them.
Can one still be vaccinated if they have a positive test for COVID-19?
Yes. But, the First question should be the type of test taken. There is currently no institution in Kenya offering Viral Culture tests of COVID19 as globally this has also not passed as the standard test. On the international scene, in China, reports have indicated that Anal swabs are better for detection of the virus through a molecular technique known as Polymerase Chain Reaction PCR, which Kenya is using (with nasal swabs, luckily) as the standard test in Government approved and accredited facilities. If the test is a rapid or ELISA test that checks on the presence of antibodies, then it may indicate that one recovered from the virus but still has the antibodies.
Then, the second question should be why not. If vaccine vials are available and the benefits outweigh the potential risks as an individual, then a Big Yes. However, it is good to note that the long-term effects of the Vaccines are not known as all are under one year since approval for human use. Some “potential side effects” that people have had questions about have proven that either we are too imaginative as a species or have watched many horror/sci-fi movies for our own good. Well being one of those vaccinated all I can say is…Let’s wait and see
What are the adverse side effects of these vaccines?
From the collected & available published data,None. None to Mild symptoms published data is available.
From the manufacturers, manufacturers associated publications, and some government reports (probably the are biased group due to potential selfish reason), Ranges from Mild-to-None. Mild side effects include but are not limited to pain at the injection site, swelling at the injection site and surrounding areas, reddening or darkening of injection area depending on one’s skin tone, fever (and night chills), headache, general body weakness, and aches, insomnia, mild nausea, vomiting & sometimes diarrhea (which might be cases of food poisoning/ age/other disease-related issues unrelated to the vaccine according to the manufacturers)
From some news reported and unpublished data; Blood clots. (Am sure the data will trickle in soon)
From people I have interacted with, ranges from none, to mild to severe similar to the reports above by the manufacturers. Mild side effects include but are not limited to pain at the injection site, swelling at the injection site and surrounding areas, reddening or darkening of injection area depending on one’s skin tone, fever (and night chills), headache, general body weakness, and aches, insomnia, and last but not least I have received reports from some vaccines of severe nausea, vomiting & severe diarrhea (which might be classified as Serious symptoms, and lastly probably a propaganda symptom from those who have received the vaccine on effects on one’s libido; both increase and decrease reported (this is not an officially recorded symptom but might true due to the role of ACE2 receptor antagonists resulting in higher libido if the antagonists are present or lower if they are absent. The Vaccine results in higher antagonism through the binding of ACE2 receptors).
Is there a vaccine that has 100% efficacy and completely safe?
Probably not, and if there is one it usually will result in disease eradication, the Smallpox way. The lack of 100% effectiveness may result actually from the pathogen mutating, immunodeficiency in certain individuals for various reasons rather than the scientific shortcomings of the virus itself.
In terms of safety, the reports from the governing agencies FDA and EMA, regulating in the US and Europe respective have remained as safe for use and in addition that the benefits outweigh the risks still. The data from the ongoing rollout of the various vaccines have ranged from “temporal stopping of the vaccination program due to blood clots (wait & see)” to “running out of vaccine vials with reported adverse effects”
Are the COVID-19 vaccines safe for people with autoimmune disorders e.g GBS CIDP variant?
Probably there are amongst the available vaccines. But lack of numbers and data on this would have the answer of Precaution and to add to it that if the Benefit outweighs the risk again, such persons including pregnant mothers can take one of the vaccines but with advice from a licensed medical practitioner who agrees to shoulder the damages (this might not happen, so I change my answer from Precaution to No). These people hopefully will benefit from herd immunity, and following MoH guidelines.
Is it safe for use by ladies who experience heavy period clots?
Precaution. The scientific analysis of the interaction of the AstraZeneca vaccine causing clots from available clinical data has ranged from no interaction to caution, to issuance of the common phrase about “benefits outweighed the risks”, to understanding of the virus vaccine and the resulting produced spike proteins being the cause or vitamin K playing a role. Some vaccines have gone ahead and taken blood-thinning drugs to “prevent” the clots though am not sure I would advise this due to the potential risk of hemorrhage.
How does a country decide to import vaccine X and not Y?
Money. Cost. Availability of enough stocks. Economic and Political ties with the producing countries e.g. India recently donated vials free of charge to the Government and all Institutions forming the COVAX global initiative and finally probable the most important advice from the Ministry of Health and the Corona Virus task force set up by our President that also advises on actions the government should take e.g the restriction of movement for 5 counties and closure of all learning institutions. AstraZeneca vaccine is also more suited for African markets looking at its storage temperatures and shelf life and this too might advise the task force’s decision.
Summary of the different COVID-19 Vaccines
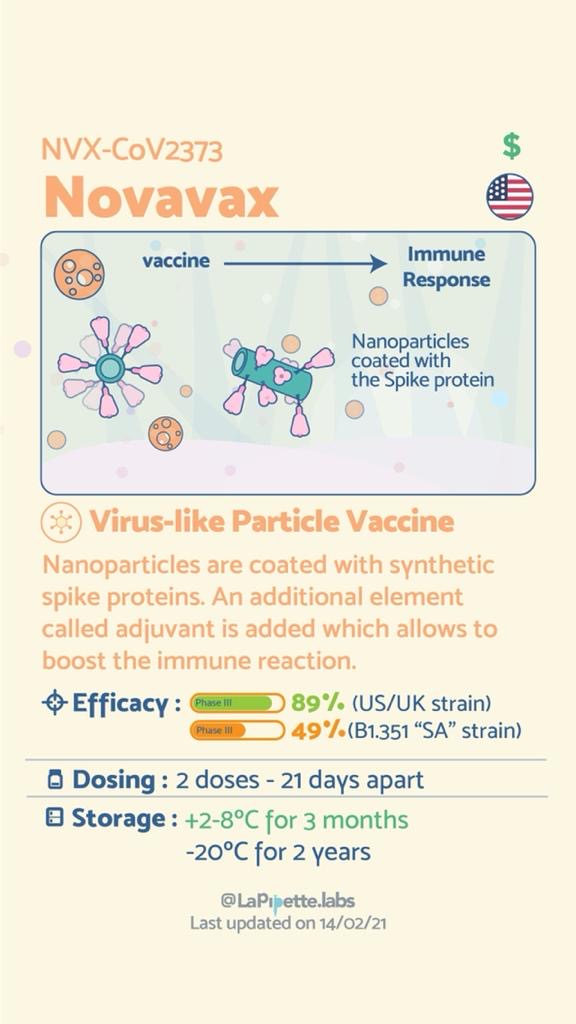
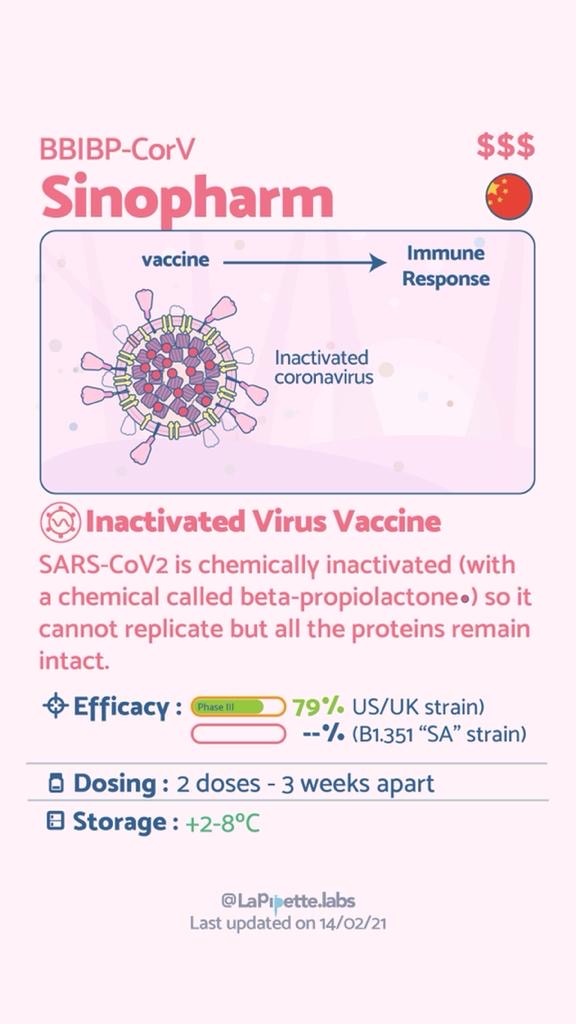
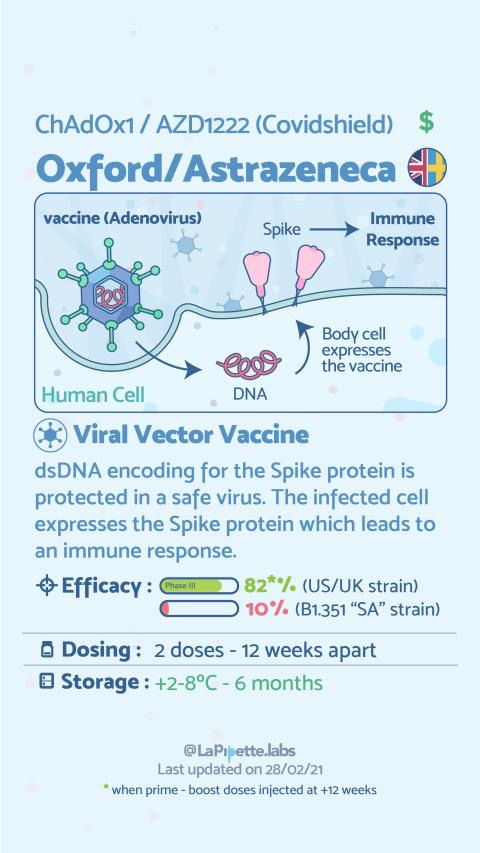
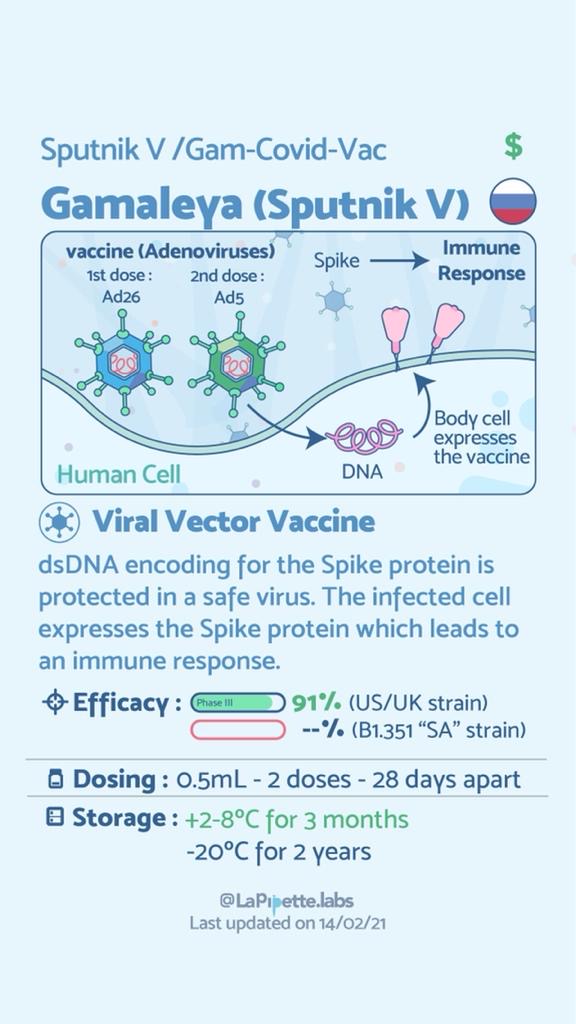
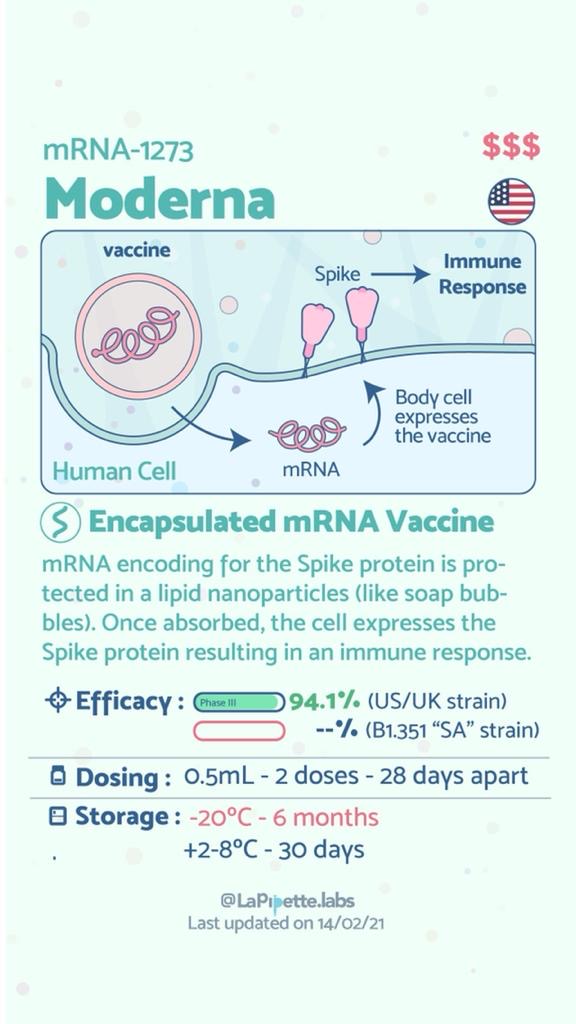
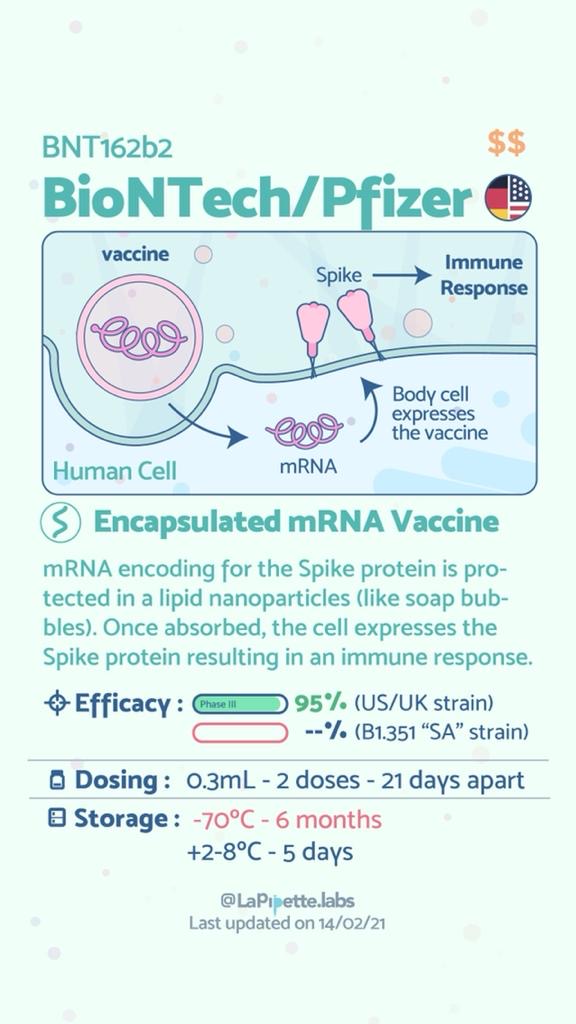
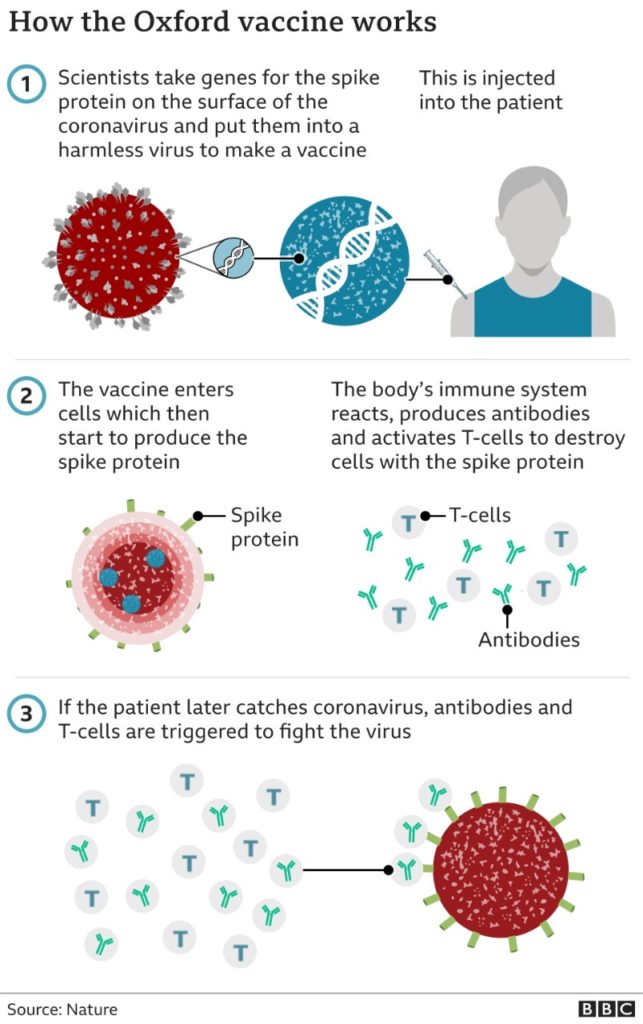
Summary of how the Oxford Vaccine works.
Contributor: Dr. Shem Mutuiri.
Tutorial Fellow in the Department of Biochemistry, Microbiology & Biochemistry.
Specialist in Medical Microbiology, Bacteriology, Virology, Immunology, Mycology, Vaccinology.
Contact: mutuiri.shem@ku.ac.ke

NO COMMENT